Ultrasound gel is an integral part of any exam or procedure conducted with the modality. For the ultrasound scanner to function, it requires an aqueous solution to be applied to the skin’s surface. Upon contact with the solution, the ultrasound transducer can then successfully send high-frequency sound waves back and forth to produce the image seen on the monitor. The technique employed by the modality is similar to the echolocation used by whales and dolphins as well as SONAR used by submarines.
While ultrasound imaging is commonly associated with prenatal scans, the modality’s use has significantly expanded in recent years. It is increasingly used across a variety of clinical areas and specialties, such as urology, cardiology, and emergency medicine. Ultrasound’s presence in interventional settings has led to the issuance of a series of infection control recommendations from regulatory bodies and professional associations.
When used in the operating theater or in exams where the risk of infection is high, ultrasound users must consider their choice of gel as well as the type of probe cover they will use. Although sometimes overlooked, ultrasound gel can harbor pathogens and lead to healthcare-associated infections. This is especially true when refillable bottles are used in lieu of sterile single-use gel packets. These refillable bottles are more difficult to clean and the consistent use of one bottle across multiple patients raises the risk of pathogenic transmission.
Ultrasound gel comes in a wide variety of colors – clear, blue, or pink – and shapes – dispenser bottle or single-use packet – but when it comes to infection control the main characteristic which clinicians should emphasize is non-sterile vs. sterile. The use of sterile gel preserves the sterility of interventional settings and works to reduce the risk of healthcare-associated infections.
When should I use sterile ultrasound gel?
Here are three different scenarios in which you’ll want to use sterile ultrasound gel:
-
If your transducer is in contact with a mucous membrane. This effectively encompasses all transrectal and transvaginal ultrasound exams.
-
If your transducer is in contact with non-intact skin.
-
During surgical ultrasound scans.
All three of these exams pose an elevated risk to patient safety. The first two fall into the second category of the Spaulding classification – semi-critical. The third scenario is classified as critical. Some organizations, such as the European Society of Radiology (ESR), have proposed a modified Spaulding classification which only includes the lowest level of risk and the highest level of risk – that is, non-critical and critical. This is mainly because the risk of pathogenic transmission remains high, even in semi-critical exams and procedures.
It is important to note that the use of a sterile probe cover, although crucial, does not remove the need for a sterile gel. Even with a probe cover, the transducer in semi-critical and critical procedures may come into contact with mucous tissue and/or blood and bodily fluids; thus, the risk of pathogen transmission remains high. There are two primary reasons against using non-sterile gel during these procedures. First, you will risk contaminating the patient with an already-contaminated gel. Second, you risk contaminating the gel container itself if the patient is high-risk or infected with a transmittable disease.
If your non-sterile gel container is already contaminated, whether from the factory or due to use on an infected patient, then the odds of pathogenic transmission are increased. For example, if your probe cover is punctured during the procedure and your gel is non-sterile and becomes infected or infects the patient, then it creates a significant gap in your infection control protocol. As an ESR study pointed out, “the risk of microscopic or macroscopic probe cover perforations necessitates consideration.” Thus, we recommend the use of a sealed single-use sterile gel packet for inside and outside of the sterile probe cover.
How to use sterile gel?
The aforementioned survey from the ESR revealed that only 14% of the audited clinicians used sterile gel for their endocavity exams. From an infection control standpoint, this finding reveals a gap in the infection control practices of ultrasound users. In critical and semi-critical procedures, clinicians must measure the risk posed by every piece of equipment used during the ultrasound procedure. This includes not only the probe itself, but also the probe cover, the ultrasound gel, the cable, and the monitor.
More importantly, 69% of the respondents reported that they simply wiped the probe at the end of an endocavity procedure. In effect, two thirds of the clinicians only performed low-level disinfection between each patient. Failure to perform high-level disinfection fosters pathogenic transmission via the ultrasound probe, and subsequently, the gel container (if single-use packets are not employed).
Healthcare-associated infections in ultrasound procedures can spread by missing just one step. Consider this scenario: an exam is performed on an infected patient, the probe cover fails and the probe becomes infected, the probe is simply wiped down but does not undergo high-level disinfection, and thus the gel bottle is infected when applied to the infected probe during the next procedure. The next patient is then infected when the probe and gel are used on him/her.
For this reason, we recommend that clinicians:
-
Use sterile ultrasound gel for endocavity procedures. We recommend the single-use packets as they are discarded and eliminate the risk of cross-contamination.
-
Use a new refill bottle on a regular basis for your non-critical, surface ultrasounds (on intact skin).
It is essential that ultrasound users take each piece of the equipment into account when performing semi-critical and critical procedures. Doing so not only improves the health and well-being of patients and staff, but it also leads to higher quality care.
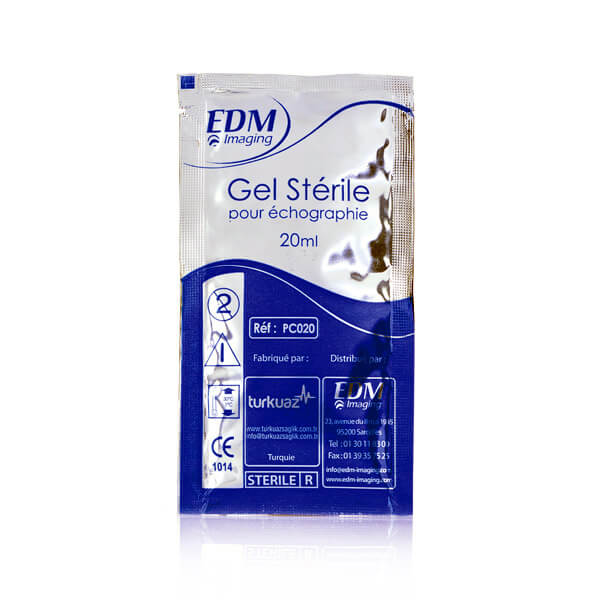 EDM Sterile Ultrasound Gel - Recommended for surgical and endocavity procedures
EDM Sterile Ultrasound Gel - Recommended for surgical and endocavity procedures Parker Aquasonic - sterile ultrasound gel
Parker Aquasonic - sterile ultrasound gel